EPITHELIAL TISSUE
Covering, Lining, and Glandular Tissue — Exploring polarity, junctions, and classification.
Overview & Origin
Definition and Embryogenesis.
What is Epithelium?
Epithelial tissue covers body surfaces, lines body cavities, and forms glands. It creates a selective barrier between the external environment and the underlying connective tissue.
Key Properties
- Cellularity: Cells are in close mutual contact (little intercellular matrix).
- Polarity: Exhibits functional and morphologic polarity (Apical vs Basal).
- Avascular: Contains no blood vessels; nutrients diffuse from connective tissue.
- Basement Membrane: Separated from connective tissue by a specialized layer.
Embryological Origin
Epithelia are unique as they derive from all three embryonic germ layers.
Classification
Lining Epithelia Types.
Classification is descriptive and based on two factors: the number of cell layers and the shape of the surface cells.

Simple Squamous
Flat, Scale-like
Function: Exchange, Lubrication.
Location: Endothelium (vessels), Mesothelium (body cavities), Alveoli.
Simple Cuboidal
Cube-shaped
Function: Secretion, Absorption.
Location: Kidney tubules, Thyroid follicles, Duct linings.
Simple Columnar
Tall, Rectangular
Function: Absorption, Secretion.
Location: Stomach, Intestine, Gallbladder.
Appears stratified because nuclei are at different levels, but all cells rest on the basement membrane. Found in the Trachea and Bronchial tree (often ciliated).
Intestinal Villus (Simple Columnar)

Surface is lined with simple columnar epithelium with microvilli (striated border).
Stomach Mucosa Histology

Example of secretory (glandular) epithelium and protective lining.
Lung Alveoli (Simple Squamous)

The thinnest possible barrier for gas exchange (simple squamous epithelium).
Kidney Tubules (Simple Cuboidal)
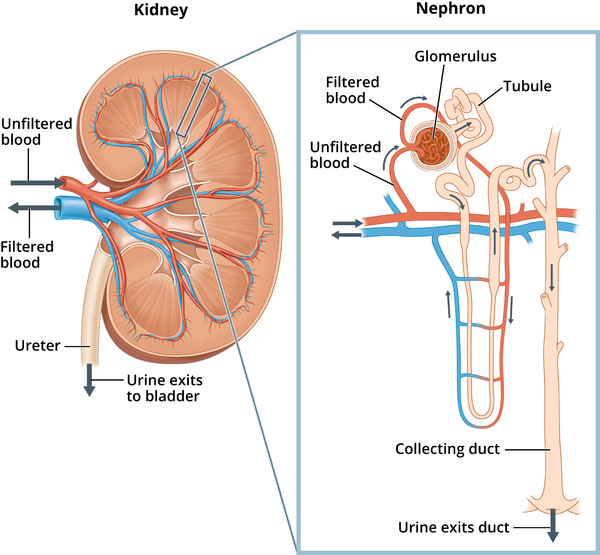
Simple cuboidal epithelium optimized for secretion and absorption in the nephron.
Liver Lobule Microstructure

Functional unit of the liver showing hepatocytes (glandular epithelium) organized around a central vein.
Classified by the shape of the cells in the superficial (apical) layer.
Stratified Squamous
Keratinized: Epidermis (dry, protective, dead surface cells).
Non-Keratinized: Oral cavity, Esophagus, Vagina (moist, protective).
Stratified Cuboidal/Columnar
Rare. Found in large ducts of exocrine glands (sweat glands, salivary glands).
Transitional (Urothelium)
Specialized for distension. Surface cells are large and dome-shaped ("umbrella cells"). Found in Bladder, Ureters.
Cell Polarity
Apical, Lateral, and Basal Domains.
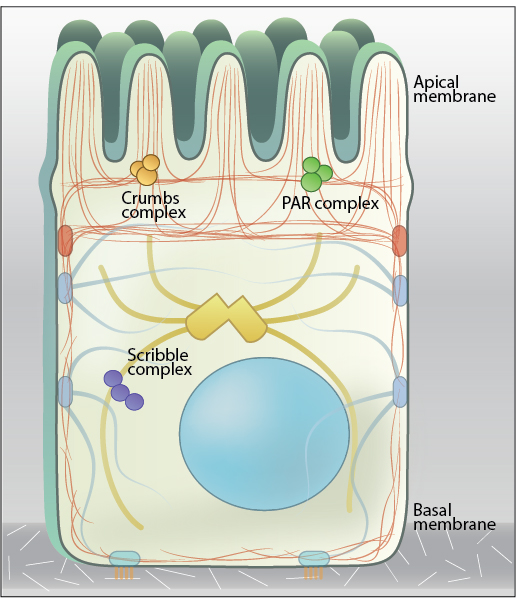
Apical Domain
Faces the lumen or external environment.
- Microvilli: Actin core. Increase absorption (e.g., Intestine).
- Stereocilia: Long microvilli. Sensory (Ear) or absorption (Epididymis).
- Cilia: Microtubule core (9+2). Movement of fluid (e.g., Trachea).
Lateral Domain
Contacts adjacent cells.
- Occluding Junctions: Tight seals.
- Anchoring Junctions: Mechanical strength.
- Gap Junctions: Communication.
Basal Domain
Rests on the basement membrane.
- Basement Membrane: Attachment & filtration.
- Hemidesmosomes: Anchor cell to matrix.
- Infoldings: Increase surface area (Ion transport).
Junctional Complex
Cell-to-Cell Adhesion and Communication.
Specialized structures in the lateral domain that adhere cells together and allow communication.
Tight Junction
Zonula Occludens
Most apical. Seals intercellular space to prevent leakage. Formed by Claudins and Occludins. Defines cell polarity.
Adherens Junction
Zonula Adherens
Below tight junctions. Encircles cell. Mediated by E-Cadherin (Ca++ dependent). Links to Actin filaments via Catenins.
Desmosome
Macula Adherens
Spot-like adhesion. Mediated by Desmogleins. Links to Intermediate Filaments (Keratin). Provides mechanical resistance.
Gap Junction
Communicating
Formed by Connexons (6 connexins). Allows passage of ions and small molecules. Coordinates activity (e.g., heart beat).
Basal Domain
Interaction with the underlying tissue.
Basement Membrane
A specialized layer of extracellular matrix that separates the epithelium from the connective tissue.
- Lamina Lucida: Electron-lucent layer just below the cell. Contains Integrins, Laminin.
- Lamina Densa: Electron-dense layer. Contains Type IV Collagen, Perlecan.
- Reticular Lamina: Produced by connective tissue. Type III (Reticular) and Type VII (Anchoring) Collagen.
Hemidesmosomes
Junctions that anchor the basal domain of the epithelial cell to the basal lamina.
- Transmembrane Protein: Integrins (α6β4).
- Intracellular Link: Plectin (connects to Intermediate Filaments).
- Extracellular Link: Laminin-332 and Collagen IV.
Cell Renewal
Turnover and Stem Cells.
Epithelia belong to the category of continuously renewing cell populations. The rate depends on the tissue.
Small Intestine
Stem cells in crypts.
Skin (Epidermis)
Stem cells in Stratum Basale.
Epithelial Review
Key Terminology.
| Term | Definition |
|---|---|
| Endothelium | Simple squamous epithelium lining blood vessels. |
| Microvilli | Apical projections containing actin; increase surface area for absorption. |
| Connexon | The functional unit of a gap junction, formed by 6 connexins. |
| Mesothelium | Simple squamous epithelium lining body cavities (Pleura, Peritoneum). |
CONNECTIVE TISSUE
The Support System — From Fat Cells to Bone Matrix.
Overview & Classification
The Matrix and Cells.
Connective tissue comprises a diverse group of cells embedded within a tissue-specific Extracellular Matrix (ECM). Unlike epithelium, cells are widely separated by this matrix.
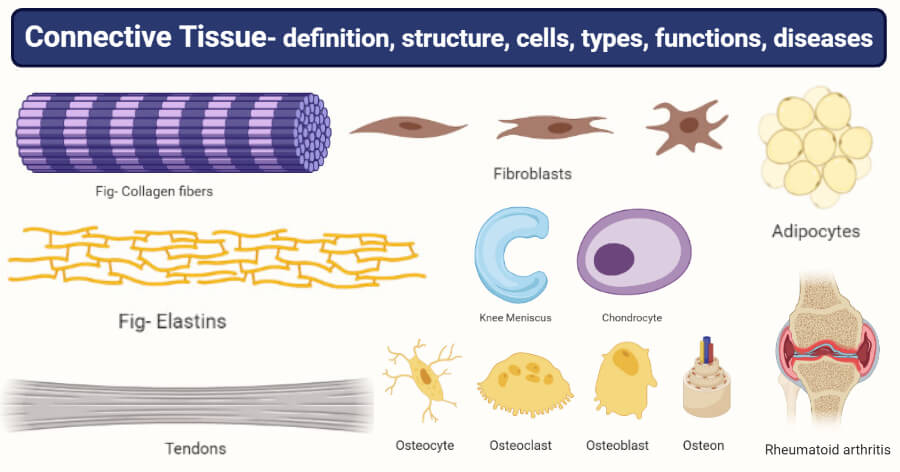
Components
- Cells: Fibroblasts, Adipocytes, Immune cells.
- Fibers: Collagen (Strength), Elastic (Stretch), Reticular (Support).
- Ground Substance: Amorphous gel (GAGs, Proteoglycans).
Classification
Adipose Tissue
Energy Storage & Insulation.
A specialized connective tissue where Adipocytes predominate. It is the largest energy reservoir in the body.

White Adipose Tissue
- Structure: Single huge lipid droplet. Nucleus flattened at periphery ("Signet ring").
- Function: Energy storage, Insulation, Cushioning.
- Location: Hypodermis, around organs (kidneys).
Brown Adipose Tissue
- Structure: Multiple small lipid droplets. Central nucleus. Many mitochondria (Iron = Brown color).
- Function: Thermogenesis (Heat production) via UCP1 (Thermogenin).
- Location: Newborns, Hibernating animals.
Cartilage
Flexible Support.
Avascular tissue. Cells (Chondrocytes) reside in lacunae within a firm gel-like matrix.

Hyaline
Glassy Appearance
- Matrix: Type II Collagen (invisible), Aggrecan, Water (60-80%).
- Location: Articular surfaces, Trachea, Nose, Fetal skeleton.
- Perichondrium: Present (except articular).
Elastic
Flexible
- Matrix: Elastic fibers + Type II Collagen.
- Location: External Ear (Pinna), Epiglottis.
- Perichondrium: Present.
Fibrocartilage
Tough
- Matrix: Thick Type I Collagen bundles + Type II.
- Location: Intervertebral discs, Pubic symphysis, Menisci.
- Perichondrium: Absent.
Bone Cells
The Architects of Bone.
Osteoblast
The Builder
Secretes organic matrix (Osteoid) including Type I Collagen. Initiates mineralization. Located on bone surfaces.
Osteocyte
The Sensor
Mature bone cell trapped in lacunae. Senses mechanical stress. Communicates via canaliculi.
Osteoclast
The Destroyer
Large, multinucleated cell derived from monocytes. Resorbs bone using acid and enzymes. Sits in Howship's lacunae.
Bone Structure
Compact vs. Spongy Bone.
Compact Bone: The Osteon
The functional unit of compact bone is the Osteon (Haversian System).
Structural Components
- Haversian CanalCentral channel containing blood vessels and nerves.
- LamellaeConcentric rings of calcified matrix.
- LacunaeSmall cavities containing Osteocytes.
- CanaliculiTiny channels connecting lacunae for nutrient exchange.
- Volkmann'sPerpendicular channels connecting Haversian canals.
Ossification
Bone Formation Types.
Intramembranous
Direct formation of bone from mesenchymal connective tissue.
- Mechanism: Mesenchymal cells differentiate directly into osteoblasts.
- Examples: Flat bones of the skull, Clavicle, Mandible.
Endochondral
Bone forms by replacing a hyaline cartilage model.
- Mechanism: Cartilage calcifies and is replaced by bone.
- Growth Plate: Epiphyseal plate allows longitudinal growth.
- Examples: Long bones (Femur, Humerus), Vertebrae.
Epiphyseal Plate Zones (Top to Bottom)
Blood & Lymph
Fluid Connective Tissues.
Blood Composition

Blood is a specialized connective tissue with a liquid ECM called Plasma.
- Erythrocytes (RBCs): Transport O2/CO2. No nucleus.
- Leukocytes (WBCs): Immune defense (Neutrophils, Lymphocytes, etc.).
- Thrombocytes (Platelets): Cell fragments for clotting.
Lymphoid Histology

Lymphoid organs (Nodes, Spleen) feature a reticular fiber framework supporting lymphocytes.
Connective Tissue Review
Key Terminology.
| Term | Definition |
|---|---|
| ECM | Extracellular Matrix; ground substance + fibers (collagen, elastic, reticular). |
| Chondrocyte | Mature cartilage cell maintaining the matrix. |
| Periosteum | Dense connective tissue covering bone (except articular surfaces). Essential for growth/repair. |
| Lacuna | A small cavity in bone or cartilage containing an osteocyte or chondrocyte. |
| Hydroxyapatite | Calcium phosphate crystals that give bone its hardness. |
NERVOUS TISSUE
Neurons, Glia, Nerves, and Electrophysiology.
Overview & Organization
The Master Control System.
Nervous tissue allows for rapid and specific communication between different areas of the body. It receives sensory input, processes information (integration), and dictates motor output. It is derived from the Ectoderm (specifically the Neural Tube and Neural Crest).
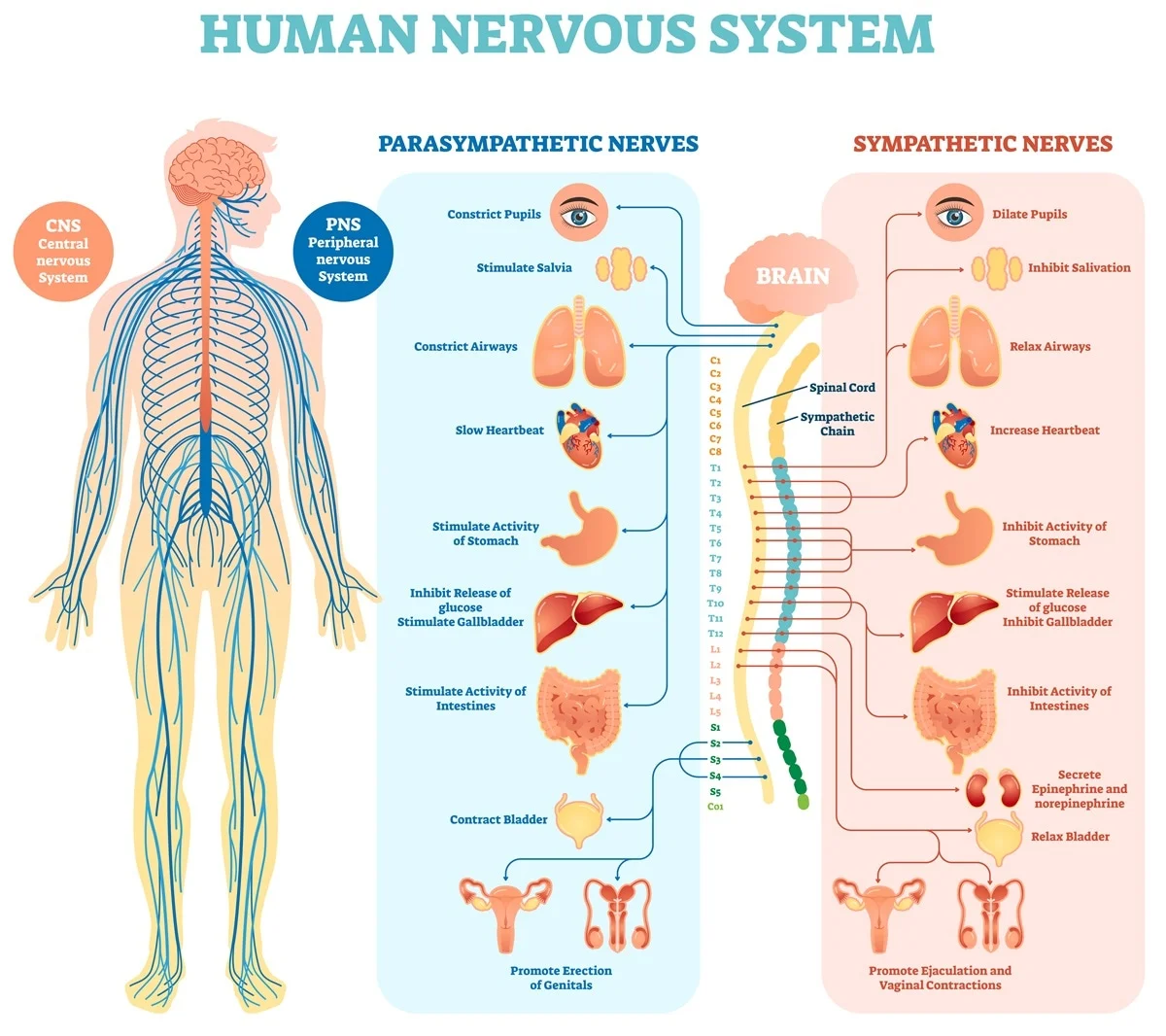
Central Nervous System
- Components: Brain (Encephalon) and Spinal Cord.
- Function: Integration and command center.
- Tissue: Contains neurons and glial cells (Astrocytes, Oligodendrocytes, Microglia, Ependymal).
- Environment: Protected by the Blood-Brain Barrier (BBB).
Peripheral Nervous System
- Components: Cranial nerves (12 pairs), Spinal nerves (31 pairs), and Ganglia.
- Function: Communication lines between the CNS and the rest of the body.
- Tissue: Neurons (Ganglia), Schwann cells, Satellite cells.
The Neuron
The Functional Unit.
Neurons are highly specialized for excitability (generating action potentials) and conductivity. They are "post-mitotic" cells (do not divide).

The metabolic center.
- Nucleus: Large, spherical, euchromatic (pale), with a prominent nucleolus ("Owl's eye" appearance).
- Nissl Bodies: Large granules of Rough ER and free ribosomes. Site of intense protein synthesis. Absent in the axon hillock.
- Cytoskeleton: Neurofilaments (intermediate filaments) for structure, Microtubules for transport.
The Input Zone.
- Short, tapering, branching extensions.
- Receive signals from other neurons.
- Dendritic Spines: Small protrusions that increase surface area for synapses. Plasticity of spines is key to memory/learning.
- Contain Nissl bodies in proximal parts.
The Conducting Zone.
- Single, long process. Arises from the Axon Hillock (trigger zone).
- Axoplasm: Contains mitochondria and microtubules but NO rough ER (no protein synthesis).
- Initial Segment: Site where Action Potential is generated. High density of Na+ channels.
- Ends in Terminal Boutons.
Axonal Transport
The Neuron's Highway System.
Since the axon contains no machinery for protein synthesis, everything must be shipped from the soma. This occurs via Microtubules acting as tracks and Motor Proteins carrying the cargo.
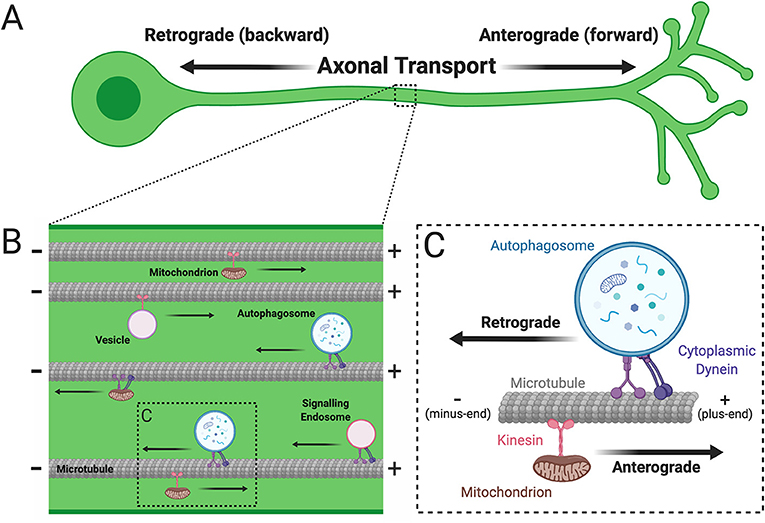
Anterograde Transport
Direction: Soma → Axon Terminal
Motor Protein: Kinesin (+ end directed)
- Fast Component: (400 mm/day). Carries mitochondria, vesicles with neurotransmitters, channel proteins.
- Slow Component: (1-5 mm/day). Carries cytoskeletal elements (actin, neurofilaments) and soluble enzymes.
Retrograde Transport
Direction: Axon Terminal → Soma
Motor Protein: Dynein (- end directed)
- Fast Only: (200 mm/day).
- Carries worn-out organelles for degradation (lysosomes).
- Carries growth factors (NGF) and signal molecules to the nucleus.
Viruses & Toxins: Rabies, Polio, Herpes simplex, and Tetanus toxin hitch a ride on Retrograde Transport to reach the CNS from the periphery.
Neuronal Classification
Structural & Functional.
Structural (Number of Processes)
- Multipolar1 Axon + Many Dendrites. The most common type. Includes motor neurons and interneurons.
- Bipolar1 Axon + 1 Dendrite. Rare. Found in special senses: Retina, Olfactory epithelium, Vestibulocochlear ganglia.
Bipolar neurons in the retina connect photoreceptors to ganglion cells.
- PseudounipolarSingle process that splits into a peripheral branch (receptor) and central branch (CNS). Sensory neurons in Dorsal Root Ganglia.
Functional (Signal Direction)
- Sensory (Afferent)Transmit impulses TO the CNS from receptors. Usually pseudounipolar.
- Motor (Efferent)Transmit impulses FROM the CNS to effectors (muscles, glands). Multipolar.
- InterneuronsConnect sensory and motor neurons within the CNS. 99% of all neurons. Responsible for integration and reflex arcs.
Neuroglia (Glia)
The Support Crew.
Glia ("Glue") are non-neuronal cells that support, nourish, and protect neurons. They are mitotic (can divide) and outnumber neurons significantly.
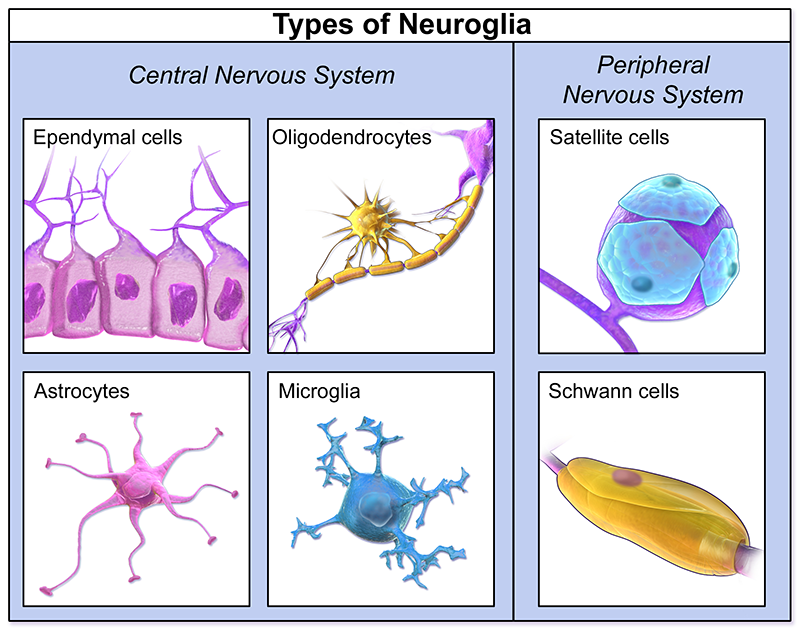
Central Nervous System (CNS)
Most abundant.
- Star-shaped with "perivascular feet".
- Form the Blood-Brain Barrier (BBB) by wrapping capillaries.
- Regulate ionic environment (K+ buffering).
- Recycle neurotransmitters (Glutamate/GABA).
- Form scar tissue (gliosis) after injury.
"Few tree branches".
- Form the Myelin Sheath in the CNS.
- One cell can wrap up to 50 different axon segments.
- Inhibits axon regeneration.
Small, thorny processes.
- Derived from Monocytes (Mesoderm), unlike others (Neuroectoderm).
- Phagocytic: The immune defense (macrophages) of the CNS.
- Activated by inflammation/injury.
Cuboidal/Columnar epithelial-like.
- Line the ventricles of the brain and central canal of the cord.
- Possess cilia and microvilli.
- Produce and circulate Cerebrospinal Fluid (CSF) (Choroid plexus).
Glia Comparison: CNS vs PNS Myelination

Oligodendrocytes (CNS) can myelinate multiple axons, while Schwann cells (PNS) wrap only one axon segment.
Peripheral Nervous System (PNS)
- Form the Myelin Sheath in the PNS.
- 1 cell = 1 axon segment.
- Vital for axon regeneration (form a regeneration tube).
- Small, flat cells surrounding neuron cell bodies in Ganglia.
- Regulate nutrient/waste exchange (similar to astrocytes).
The Myelin Sheath
Insulation for Speed.
Myelin is a whitish, fatty (lipid-rich) substance formed by concentric wrapping of glial plasma membranes around axons. It acts as an electrical insulator, preventing charge leakage.
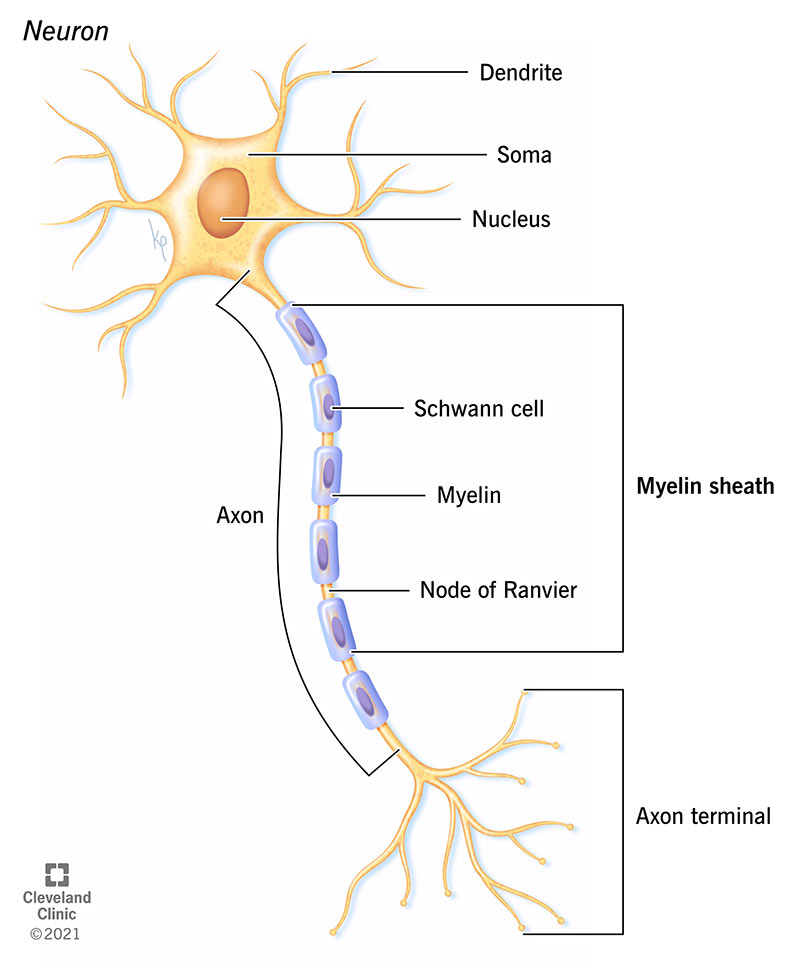
Structure
- Composition: 80% Lipid, 20% Protein.
- Nodes of Ranvier: Gaps in the sheath (approx 1 μm wide). High density of Voltage-Gated Na+ channels. Site of action potential regeneration.
- Internodes: The insulated segments between nodes.
Saltatory Conduction
In myelinated axons, the action potential "jumps" (saltare) from node to node. This is:
- Faster: Up to 120 m/s (vs 1-2 m/s in unmyelinated).
- Energy Efficient: Na+/K+ pumps only need to work at the nodes.
- Multiple Sclerosis (MS): Autoimmune destruction of Oligodendrocytes (CNS). Leads to visual disturbances, muscle weakness, speech issues.
- Guillain-Barré Syndrome: Autoimmune attack on Schwann cells (PNS). Ascending paralysis.
Peripheral Nerve Structure
Organization of Nerve Fibers.
A "nerve" in the PNS is a cable-like bundle of axons enclosed by successive layers of connective tissue, similar to skeletal muscle organization.

Dense irregular connective tissue wrapping the entire nerve. Provides tensile strength and protection.
Specialized connective tissue wrapping fascicles (bundles of axons).
Crucial: Forms the Blood-Nerve Barrier via tight junctions.
Loose delicate connective tissue (reticular fibers) wrapping individual fibers (outside the myelin/Schwann cell). Electrically isolates fibers.
Electrophysiology
The Spark of Life.
1. Resting Membrane Potential (-70 mV)
The neuron is polarized (inside is negative relative to outside).
- Na+/K+ Pump: Actively pumps 3 Na+ OUT and 2 K+ IN (electrogenic). Creates gradients.
- Leak Channels: Membrane is much more permeable to K+ than Na+. K+ leaks out down its gradient, leaving negative anions behind. This is the primary determinant of RMP.
2. The Action Potential

An "All-or-None" electrical event propagating down the axon.
Tetrodotoxin (Pufferfish): Blocks Voltage-gated Na+ channels. Prevents depolarization -> Paralysis.
Local Anesthetics (Lidocaine): Also block Na+ channels, stopping pain signals.
The Synapse
Signal Transmission.
The specialized junction where a neuron communicates with another cell. Most are Chemical Synapses.

Transmission Steps:
- Action potential reaches the Presynaptic Terminal.
- Voltage-gated Calcium (Ca2+) channels open. Ca2+ enters.
- Ca2+ triggers synaptic vesicles to fuse with membrane (Exocytosis).
- Neurotransmitter is released into the Synaptic Cleft (20-30 nm gap).
- Neurotransmitter binds to receptors on the Postsynaptic Membrane.
- Ion channels open, causing a Graded Potential.
- Neurotransmitter is removed (reuptake, enzymatic degradation, or diffusion).
Opens Na+ channels. Depolarizes postsynaptic cell. Increases chance of AP. (e.g., Glutamate, ACh).
Opens K+ or Cl- channels. Hyperpolarizes postsynaptic cell. Decreases chance of AP. (e.g., GABA, Glycine).
MUSCLE TISSUE
From the Angstrom-scale sliding filaments to the macroscopic generation of force. Skeletal, Cardiac, and Smooth muscle biology.
Classification of Muscle Tissue
The contractile machinery of the body. Muscle tissue converts chemical energy (ATP) into mechanical energy (Force).
Skeletal
Voluntary • Striated
- Morphology: Long, cylindrical, multinucleated syncytium.
- Nuclei: Peripheral (under sarcolemma).
- Regeneration: Satellite cells (limited).
- Function: Posture, movement, heat.
Cardiac
Involuntary • Striated
- Morphology: Branched, short cylinders.
- Nuclei: Single, central (halo of glycogen).
- Junctions: Intercalated Discs.
- Regeneration: None (fibrosis).
Smooth
Involuntary • Non-Striated
- Morphology: Fusiform (spindle).
- Nuclei: Single, central (corkscrew).
- Structure: Dense bodies (no sarcomeres).
- Regeneration: Active (mitosis).
SKELETAL MUSCLE
Structure, Organization, and the Molecular Engine.
Connective Tissue Hierarchy
The transmission of force relies on the continuity of connective tissue layers, which merge to form tendons (Myotendinous Junction). These layers also convey neurovascular bundles.

- EpimysiumDense irregular connective tissue surrounding the entire muscle.
- PerimysiumConnective tissue surrounding fascicles (bundles of fibers). Main route for larger nerves/vessels.
- EndomysiumDelicate reticular fibers (Type III Collagen) surrounding individual fibers. Supports capillaries and synaptic terminals.
Microanatomy & Organelles

- Sarcolemma: Plasma membrane. Forms T-Tubules.
- Sarcoplasm: Cytoplasm. Contains Myoglobin (O2 storage) and Glycogen.
- Sarcoplasmic Reticulum (SR): Specialized SER. Forms Terminal Cisternae. Stores Ca2+ (bound to Calsequestrin).
- Triad: 1 T-Tubule + 2 Terminal Cisternae. Located at the A-I Junction. Crucial for E-C coupling.
- Satellite Cells: Stem cells located between the basal lamina and sarcolemma. Responsible for limited regeneration.
The Sarcomere
Functional Unit: Z-line to Z-line (2.5 μm relaxed)
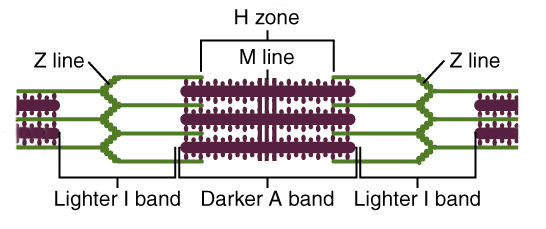
Note: During contraction, the I-Band and H-Zone shorten. The A-Band length remains constant.
Excitation-Contraction Coupling
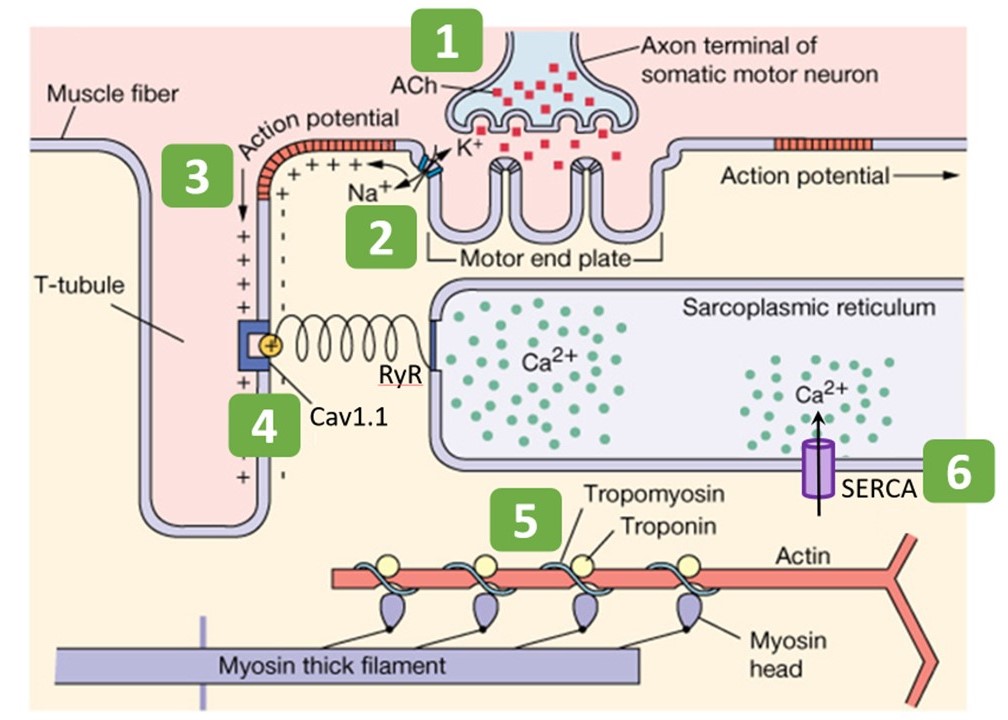
The Sequence of Events
- Neuromuscular Transmission: Action Potential travels down motor neuron axon -> Voltage-gated Ca2+ channels open -> Ca2+ entry triggers exocytosis of synaptic vesicles containing Acetylcholine (ACh).
- Sarcolemma Depolarization: ACh diffuses across synaptic cleft -> Binds Nicotinic ACh receptors on the motor end plate -> Na+ influx generates End Plate Potential (EPP) -> Action Potential sweeps across sarcolemma. ACh is degraded by Acetylcholinesterase.
- T-Tubule Transmission: AP dives into T-tubules. The voltage change is sensed by Dihydropyridine Receptors (DHPR) (L-type Ca2+ channels).
- Mechanical Coupling: In skeletal muscle, DHPR is mechanically coupled to the Ryanodine Receptor (RyR1) on the SR. The conformational change of DHPR physically pulls RyR1 open (like a cork).
- Calcium Flood: Ca2+ rushes out of SR into sarcoplasm (increasing from 0.1μM to 10μM).
- Cross-Bridge Activation: Ca2+ binds to Troponin C -> Troponin complex changes shape -> Moves Tropomyosinaway from actin active sites -> Myosin binds Actin -> Power Stroke (ATP hydrolysis).
- Relaxation: SERCA pumps (Ca2+-ATPase) actively pump Ca2+ back into SR. Calsequestrin binds Ca2+ in SR to reduce free Ca2+ gradient.
Rigor Mortis
After death, ATP production stops.
1. Without ATP, SERCA pumps fail -> Ca2+ leaks from SR -> Binding sites exposed -> Cross-bridges form.
2. Crucially: ATP is required for the Myosin head to detach from Actin. Without ATP, myosin stays locked (Rigor complexes).
3. Muscles remain stiff until proteolytic enzymes break down the sarcomeres (15-24 hours).
Duchenne Muscular Dystrophy (DMD)
Defect: Mutation in Dystrophin gene (X-linked).
Function: Dystrophin links actin cytoskeleton to ECM (Dystroglycan complex), stabilizing the membrane during contraction.
Pathology:Membrane tears -> Ca2+ influx -> Protease activation -> Necrosis. Muscle is replaced by fat (Pseudohypertrophy).
Innervation & Proprioception
The Motor Unit
Defined as one alpha-motor neuron and all the muscle fibers it innervates. Fibers of a unit are scattered throughout the muscle.
- Fine Control: 1 neuron : 5-10 fibers (Eye, Finger).
- Coarse Control: 1 neuron : 1000+ fibers (Back, Thigh).
- Recruitment: Small units (Type I) are recruited first; larger units (Type II) recruited for more force (Size Principle).
Myasthenia Gravis
Autoimmune disease where antibodies block/destroy Nicotinic ACh receptors at the NMJ. Causes progressive weakness (ptosis, diplopia). Treatment: AChE inhibitors.
The Muscle Spindle (Stretch Sensor)
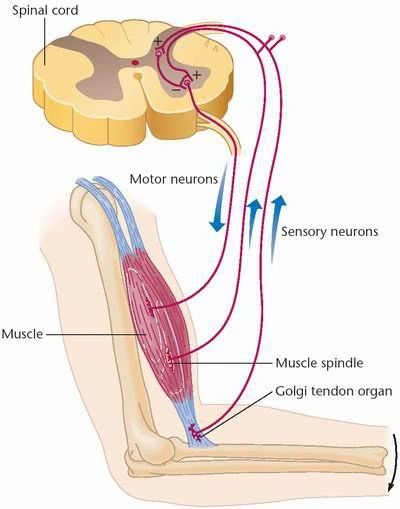
Detects changes in length. Activates stretch reflex.
CARDIAC MUSCLE
Endurance, Automatism, and Intercalated Discs.
Structural Unique Features
- Cardiocytes: Short, cylindrical, and branched cells (Y-shaped). This allows for a complex 3D network.
- Nucleus: Typically single, pale, large, and centrally located. Occasionally binucleated.
- Metabolism: Mitochondria are huge (40% vol). Depends on aerobic metabolism (Fatty acids > Glucose). Very little glycogen compared to skeletal.
- Diads: T-tubules are wider and located at the Z-line (not A-I junction). They associate with only one terminal cisterna (Diad).
Endocrine Function
Atrial cells contain specific granules producing Atrial Natriuretic Peptide (ANP).
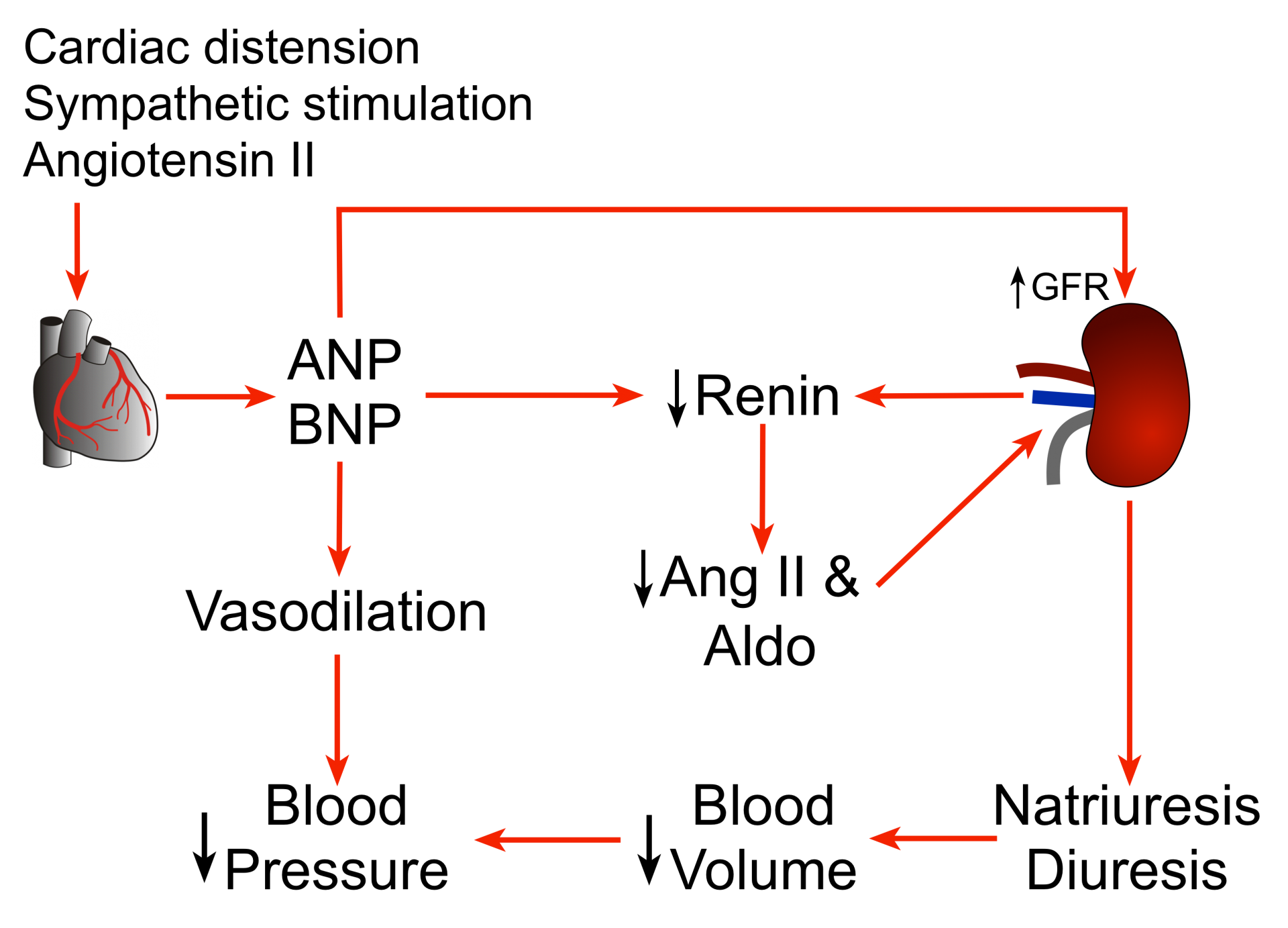
Action: Acts on Kidney -> Increases Na+ and H2O excretion -> Decreases Blood Volume/Pressure.
The Intercalated Disc
The structural and electrical bridge between cardiomyocytes.

Perpendicular to contraction. Holds cells together mechanically.
Parallel to contraction. Low electrical resistance for AP propagation.
Conduction & Physiology
CICR Mechanism
- Trigger: Action Potential opens L-type Ca2+ channels (DHPR) in T-tubule.
- Influx: Extracellular Ca2+ enters. (Note: Not enough for contraction by itself).
- Activation: This "Trigger Calcium" binds to RyR2 on the SR.
- Release: RyR2 opens (Calcium-Induced Calcium Release). Massive Ca2+ flood.
- Regulation: Sympathetic system phosphorylates Phospholamban on the SR, speeding up SERCA pumps -> Faster relaxation (Lusitropy) and stronger contraction (Inotropy).
Purkinje Fibers (Conduction)
Modified cardiomyocytes specialized for electrical conduction, not contraction.
- Structure: Larger diameter than ordinary myocytes. Paler cytoplasm (High Glycogen, few myofibrils).
- Location: Subendocardial layer.
- Function: Rapid transmission of impulse from AV bundle to ventricular myocardium.
- Junctions: Massive number of gap junctions.
SMOOTH MUSCLE
Visceral Control. Plasticity, Economy, and the Latch State.
Ultrastructure
- Shape: Fusiform (spindle). Tapered ends overlap.
- Filaments: No sarcomeres. Actin and Myosin criss-cross the cell, anchored to Dense Bodies (functional Z-lines).
- Dense Bodies: Located in cytoplasm and plasma membrane. Contain α-actinin. Anchor Intermediate Filaments (Desmin/Vimentin) which transmit force.
- Caveolae: Membrane invaginations rich in Ca2+ channels. Analogous to T-tubules but less organized.
- Regeneration: Unique among muscles. Retains mitotic activity. Example: Uterus during pregnancy (Hypertrophy + Hyperplasia). Pericytes can also differentiate into smooth muscle.
Innervation: "En Passant"
No structured Neuromuscular Junction.
Autonomic axons have swellings (varicosities) containing neurotransmitters (NE, ACh). They release transmitter into the general vicinity of the muscle sheet (Diffuse Junction).
- Single-Unit (Visceral): Cells connected by Gap Junctions. Contract as a wave (Gut, Uterus). Pacemaker activity.
- Multi-Unit: Independent cells. Rich nerve supply. Precise control (Iris, Vas deferens).
Contraction Mechanism
The "Corkscrew" Shortening.
Molecular Regulation
The Calmodulin-MLCK Pathway
Smooth muscle lacks Troponin. It relies on Myosin-based regulation.
- Stimulation: Neural (ANS), Hormonal (Oxytocin, Histamine), or Stretch.
- Ca2+ Increase: Influx + SR Release (IP3 pathway).
- Calmodulin: 4 Ca2+ ions bind to Calmodulin (CaM).
- Activation: Ca2+-CaM complex activates Myosin Light Chain Kinase (MLCK).
- Phosphorylation: MLCK phosphorylates the Myosin Light Chain. This unfolds the myosin tail and activates the ATPase.
- Contraction: Cycling occurs (slower ATP hydrolysis than skeletal).
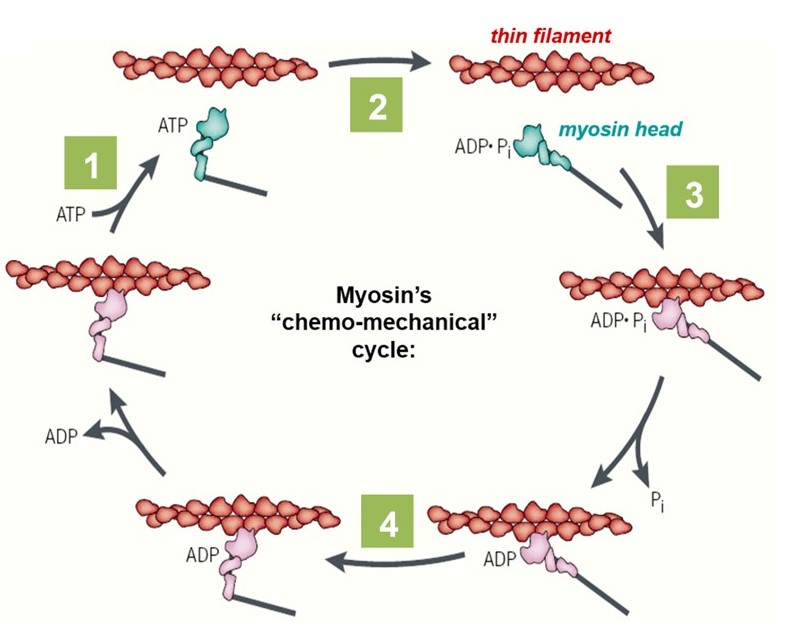
Unique to smooth muscle. Once contracted, Myosin Phosphatase can remove the phosphate while myosin is still attached to actin. This creates a "Latch Bridge" that maintains tension with near-zero ATP consumption. Essential for tonic contraction (sphincters, blood vessels).
Asthma & Bronchoconstriction
Bronchioles have smooth muscle walls. In asthma, mast cells release Histamine/Leukotrienes. These bind GPCRs on smooth muscle -> IP3 pathway -> Ca2+ Release -> Sustained contraction (Bronchospasm).
Treatment: β2-Agonists (Albuterol) increase cAMP -> PKA -> Inhibits MLCK -> Relaxation.
The Grand Comparison
Summary of key histological and physiological differences.
| Feature | Skeletal | Cardiac | Smooth |
|---|---|---|---|
| Cell Shape | Long Cylindrical | Branched Cylindrical | Fusiform (Spindle) |
| Nucleus | Many, Peripheral | 1-2, Central | 1, Central |
| Striations | Yes (Sarcomeres) | Yes (Sarcomeres) | No (Dense Bodies) |
| T-Tubules | Yes (Triads at A-I) | Yes (Diads at Z-line) | No (Caveolae) |
| Ca2+ Sensor | Troponin C | Troponin C | Calmodulin |
| Ca2+ Source | SR Only | Extracellular (Trigger) + SR | Extracellular + SR |
| Control | Voluntary (Somatic) | Involuntary (Autonomic) | Involuntary (Autonomic) |
| Regeneration | Limited (Satellite Cells) | None (Fibrosis) | High (Mitosis/Pericytes) |
| Secretory | No | Yes (ANP) | Yes (ECM molecules) |
Module Final Exam
Test your mastery of Epithelial, Connective, Nervous, and Muscle tissue with this comprehensive 40-question doctoral-level assessment.